Ultrafast Molecular Photoswitch
An international collaboration led by researchers from the Kansas State University Physics Department and the Oxford University Chemistry Department has studied ultrafast processes in the molecular photoswitch quadricyclane, published recently in Nature Chemistry.
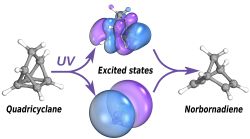
Quadricyclane and norbornadiene are two isomers, that is molecules with the same chemical formula but different geometry, which interconvert upon absorption of ultraviolet light. Quadricyclane is the higher-energy isomer and can be used to store solar energy, which is then released by a catalytic process as heat. So-called molecular solar thermal (MOST) devices exploit such isomer pairs to, for instance, warm houses by absorbing energy from the sun during the day and then releasing it at night.
The experiment, led by Kansas State Physics professor Daniel Rolles and his graduate student Kurtis Borne, used ultrashort light pulses produced by the FERMI free-electron laser facility in Trieste, Italy, to track the conversion between the two isomers on a timescale of femtoseconds. The experimental results were interpreted with the help of state-of-the-art computer simulations done in Oxford. The combined study discovered faster than previously identified pathways leading to the conversion, which may help designing new ways of controlling the outcome and efficiency of this important class of photoreactions.
See also our press release via the K-State Physics Department.
Recently Published Papers:
-
Rotation in attosecond vibronic coherence spectroscopy for molecules
Chi-Hong Yuen, Chii-Dong Lin
Commun. Phys. 7, 115 (2024) -
Exploring the ultrafast and isomer-dependent photodissociation of iodothiophenes via site-selective ionization
Weronika O. Razmus, Surjendu Bhattacharyya, Huynh V. S. Lam, Daniel Rolles, Artem Rudenko, Anbu Venkatachalam, et al.
Phys. Chem. Chem. Phys., 26, 12725 (2024)
This Week at JRM
Week of 28 April 2024
| Mon | 1:30 pm | Nuts & Bolts |
| News & status (Hybrid, CW119) | ||
| Tue | ||
| Wed | 1:30 pm | AMO Seminar |
| Tu Nguyen, KSU (CW144/145) | ||
| Thu | 9:00 am | PhD Defense |
| Tomthin Wangjam, KSU (CW119) | ||
| Fri | ||
What's New?
JRM research was featured in the Science magazine "In The Pipeline" blog by Derek Lowe this March. The piece is titled "Very Small and Very Fast" and marvels at how the work "illustrates being able to determine molecular species on a very small scale and (even more importantly) on an extremely short time scale." The original paper appears in the Journal of the American Chemical Society.